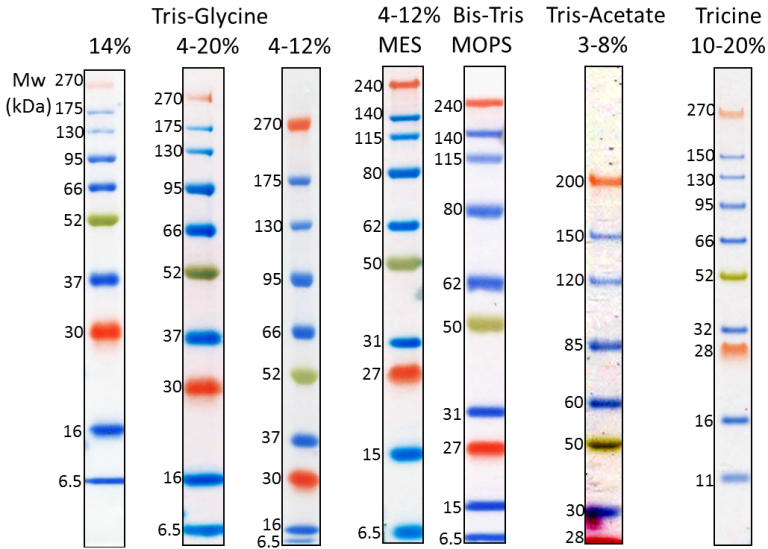
Although it is impossible to define "precision" for molecular weight of proteins in SDS-PAGE, we did compare the migration pattern of pre-stained markers with unstained protein marker (Invitrogen MARK12) for calibration. Therefore, in the product description, we suggest our users to calibrate the MW against their interested proteins. We did compare the migration patterns of SMOBIO’s Protein Markers with other brands, and we concluded that it was difficult to define “precision” due to the reasons mentioned above. For example, a protein which is highly hydrophilic might show a particular higher position in the SDS-PAGE analysis when compared to a hydrophobic one. It is known that the analysis of protein size by an SDS-PAGE is only for “estimation” because of the intrinsic variation of amino acid composition in all proteins including stained and non-stained ones. Usually, pre-stained marker is written on “estimated molecular weight” for caution. MOPS, and MES buffer) and after Western blotting to nitrocellulose membrane. Under suggested conditions, PM5200 ExcelBand™ 3-color Pre-Stained Protein Ladder Broad Range resolves 15 major bands in SDS-PAGE ( Tris-Glycine, Three reference bands - 75 kDa (red), 40 kDa (green), and 20 kDa (blue)Īpproximately 0.1~0.4 mg/ml of each protein in the buffer (20 mM Tris-phosphate (pH 7.5 at 25☌), 2% SDS, 0.2 mM DTT, 3.6 M urea, and 15% (v/v) glycerol). Ready-to-use - Premixed with a loading buffer for direct loading, no need to boil. The PM5200 3-color Pre-Stained Protein Ladder Broad Range is designed for monitoring protein separation during SDS-polyacrylamide gel electrophoresis, verification of Western transfer efficiency on membranes (PVDF, nylon, or nitrocellulose) and for approximating the size of proteins. Proteins are covalently coupled with different chromophores for easy identification of bands, with three reference proteins carrying enhanced intensity corresponding to a blue band at 20 kDa, green at 40 kDa, and red at 75 kDa, respectively, as separated on SDS-PAGE (Tris-Glycine buffer). 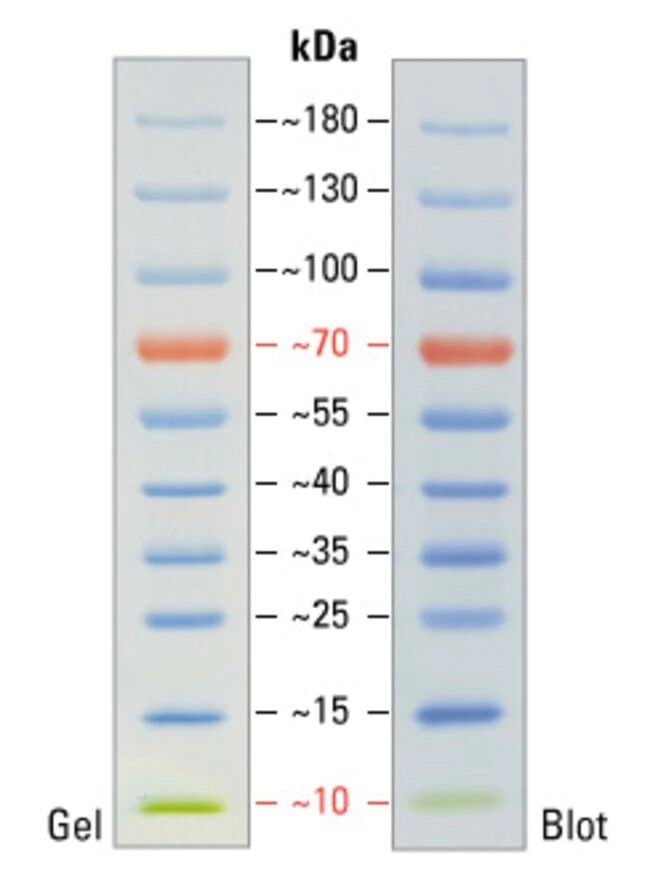
We will look at how to do this in the next post.The PM5200 3-color Pre-Stained Protein Ladder Broad Range is a ready-to-use three-color protein standard with 15 pre-stained proteins covering a wide range of molecular weights from 5 to 245 kDa in Tris-Glycine Buffer (3.5 to 235 kDa in Bis-Tris (MOPS) buffer and Bis-Tris (MES) buffer). However, you may want to use the R f values from your western blot to determine the molecular weight of your protein of interest. As the top value will always be larger than the bottom value, your result will be less than 1.Īnd that’s it! As you can see, calculating R f is relatively straightforward. Use the formula above to calculate the R f value. Once you have measured this once, you can use it to calculate Rf values of different proteins in your sample.ģ. how far the dye has moved from the top of the gel. Measure the migration distance of the dye front i.e. Make sure you know which way round your image should be, so that you measure the correct distance! For example, if the anode (+) was at the bottom of the gel, the negatively- charged proteins will have moved from the top towards the bottom of the gel.Ģ. Measure the distance your protein of interest has travelled, in millimetres. To work out the R f value by hand (rather than using software), follow these simple steps:ġ. In western blot, calculating the R f value is useful because it allows the molecular weight of a protein to be determined. The term originates in chromatography, where it was observed that a particular compound will always travel the same distance in a particular solvent, as long as conditions are constant. 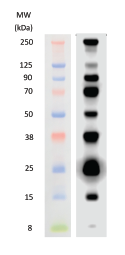
The R f (retardation factor) value is the ratio of the distance moved by the solute to the distance moved by the solvent. The final result is – hopefully – a clear image with defined bands showing the target protein. Then a secondary antibody that can be visualised (through staining or immunofluorescence) is added. The protein of interest is marked by washing the membrane with a solution containing a primary antibody. The proteins are then transferred from the gel to a membrane (the “blotting” part). Essentially, western blotting allows a mixture of proteins to be separated using gel electrophoresis. Western blot is a common tool for identifying, quantifying and determining the size of specific proteins. How to calculate the R f value of a protein from western blot
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
